Nephrology
Citation Information: J Clin Invest. 2016. https://doi.org/10.1172/JCI75079.
Abstract
Renal preglomerular arterioles regulate vascular tone to ensure a large pressure gradient over short distances, a function that is extremely important for maintaining renal microcirculation. Regulation of renal microvascular tone is impaired in salt-sensitive (SS) hypertension–induced nephropathy, but the molecular mechanisms contributing to this impairment remain elusive. Here, we assessed the contribution of the SH2 adaptor protein p66Shc (encoded by
Authors
Bradley Miller, Oleg Palygin, Victoriya A. Rufanova, Andrew Chong, Jozef Lazar, Howard J. Jacob, David Mattson, Richard J. Roman, Jan M. Williams, Allen W. Cowley Jr., Aron M. Geurts, Alexander Staruschenko, John D. Imig, Andrey Sorokin
Citation Information: J Clin Invest. 2016. https://doi.org/10.1172/JCI85265.
Abstract
Membranous nephropathy (MN) is the most common cause of nephrotic syndrome in adults, and one-third of patients develop end-stage renal disease (ESRD). Circulating autoantibodies against the podocyte surface antigens phospholipase A2 receptor 1 (PLA2R1) and the recently identified thrombospondin type 1 domain–containing 7A (THSD7A) are assumed to cause the disease in the majority of patients. The pathogenicity of these antibodies, however, has not been directly proven. Here, we have reported the analysis and characterization of a male patient with THSD7A-associated MN who progressed to ESRD and subsequently underwent renal transplantation. MN rapidly recurred after transplantation. Enhanced staining for THSD7A was observed in the kidney allograft, and detectable anti-THSD7A antibodies were present in the serum before and after transplantation, suggesting that these antibodies induced a recurrence of MN in the renal transplant. In contrast to PLA2R1, THSD7A was expressed on both human and murine podocytes, enabling the evaluation of whether anti-THSD7A antibodies cause MN in mice. We demonstrated that human anti-THSD7A antibodies specifically bind to murine THSD7A on podocyte foot processes, induce proteinuria, and initiate a histopathological pattern that is typical of MN. Furthermore, anti-THSD7A antibodies induced marked cytoskeletal rearrangement in primary murine glomerular epithelial cells as well as in human embryonic kidney 293 cells. Our findings support a causative role of anti-THSD7A antibodies in the development of MN.
Authors
Nicola M. Tomas, Elion Hoxha, Anna T. Reinicke, Lars Fester, Udo Helmchen, Jens Gerth, Friederike Bachmann, Klemens Budde, Friedrich Koch-Nolte, Gunther Zahner, Gabriele Rune, Gerard Lambeau, Catherine Meyer-Schwesinger, Rolf A.K. Stahl
Citation Information: J Clin Invest. 2016. https://doi.org/10.1172/JCI83942.
Abstract
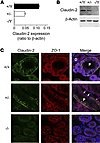
Efficient oxygen utilization in the kidney may be supported by paracellular epithelial transport, a form of passive diffusion that is driven by preexisting transepithelial electrochemical gradients. Claudins are tight-junction transmembrane proteins that act as paracellular ion channels in epithelial cells. In the proximal tubule (PT) of the kidney, claudin-2 mediates paracellular sodium reabsorption. Here, we used murine models to investigate the role of claudin-2 in maintaining energy efficiency in the kidney. We found that claudin-2–null mice conserve sodium to the same extent as WT mice, even during profound dietary sodium depletion, as a result of the upregulation of transcellular Na-K-2Cl transport activity in the thick ascending limb of Henle. We hypothesized that shifting sodium transport to transcellular pathways would lead to increased whole-kidney oxygen consumption. Indeed, compared with control animals, oxygen consumption in the kidneys of claudin-2–null mice was markedly increased, resulting in medullary hypoxia. Furthermore, tubular injury in kidneys subjected to bilateral renal ischemia-reperfusion injury was more severe in the absence of claudin-2. Our results indicate that paracellular transport in the PT is required for efficient utilization of oxygen in the service of sodium transport. We speculate that paracellular permeability may have evolved as a general strategy in epithelial tissues to maximize energy efficiency.
Authors
Lei Pei, Glenn Solis, Mien T.X. Nguyen, Nikhil Kamat, Lynn Magenheimer, Min Zhuo, Jiahua Li, Joshua Curry, Alicia A. McDonough, Timothy A. Fields, William J. Welch, Alan S.L. Yu
Citation Information: J Clin Invest. 2016. https://doi.org/10.1172/JCI83000.
Abstract
Physiochemical stress induces tissue injury as a result of the detection of abnormal molecular patterns by sensory molecules of the innate immune system. Here, we have described how the recently discovered C-type lectin collectin-11 (CL-11, also known as CL-K1 and encoded by
Authors
Conrad A. Farrar, David Tran, Ke Li, Weiju Wu, Qi Peng, Wilhelm Schwaeble, Wuding Zhou, Steven H. Sacks
Citation Information: J Clin Invest. 2016. https://doi.org/10.1172/JCI84712.
Abstract
Experimentally, females show an improved ability to recover from ischemia-reperfusion injury (IRI) compared with males; however, this sex-dependent response is less established in humans. Here, we developed a series of murine renal ischemia and transplant models to investigate sex-specific effects on recovery after IRI. We found that IRI tolerance is profoundly increased in female mice compared with that observed in male mice and discovered an intermediate phenotype after neutering of either sex. Transplantation of adult kidneys from either sex into a recipient of the opposite sex followed by ischemia at a remote time resulted in ischemia recovery that reflected the sex of the recipient, not the donor, revealing that the host sex determines recovery. Likewise, renal IRI was exacerbated in female estrogen receptor α–KO mice, while female mice receiving supplemental estrogen before ischemia were protected. We examined data from the United Network for Organ Sharing (UNOS) to determine whether there is an association between sex and delayed graft function (DGF) in patients who received deceased donor renal transplants. A multivariable logistic regression analysis determined that there was a greater association with DGF in male recipients than in female recipients. Together, our results demonstrate that sex affects renal IRI tolerance in mice and humans and indicate that estrogen administration has potential as a therapeutic intervention to clinically improve ischemia tolerance.
Authors
David D. Aufhauser Jr., Zhonglin Wang, Douglas R. Murken, Tricia R. Bhatti, Yanfeng Wang, Guanghui Ge, Robert R. Redfield III, Peter L. Abt, Liqing Wang, Nikolaos Svoronos, Arwin Thomasson, Peter P. Reese, Wayne W. Hancock, Matthew H. Levine
Citation Information: J Clin Invest. 2016. https://doi.org/10.1172/JCI83551.
Abstract
Renal peritubular interstitial fibroblast-like cells are critical for adult erythropoiesis, as they are the main source of erythropoietin (EPO). Hypoxia-inducible factor 2 (HIF-2) controls EPO synthesis in the kidney and liver and is regulated by prolyl-4-hydroxylase domain (PHD) dioxygenases PHD1, PHD2, and PHD3, which function as cellular oxygen sensors. Renal interstitial cells with EPO-producing capacity are poorly characterized, and the role of the PHD/HIF-2 axis in renal EPO-producing cell (REPC) plasticity is unclear. Here we targeted the PHD/HIF-2/EPO axis in FOXD1 stroma-derived renal interstitial cells and examined the role of individual PHDs in REPC pool size regulation and renal EPO output. Renal interstitial cells with EPO-producing capacity were entirely derived from FOXD1-expressing stroma, and
Authors
Hanako Kobayashi, Qingdu Liu, Thomas C. Binns, Andres A. Urrutia, Olena Davidoff, Pinelopi P. Kapitsinou, Andrew S. Pfaff, Hannes Olauson, Annika Wernerson, Agnes B. Fogo, Guo-Hua Fong, Kenneth W. Gross, Volker H. Haase
Citation Information: J Clin Invest. 2016. https://doi.org/10.1172/JCI83658.
Abstract
The nervous and immune systems interact in complex ways to maintain homeostasis and respond to stress or injury, and rapid nerve conduction can provide instantaneous input for modulating inflammation. The inflammatory reflex referred to as the cholinergic antiinflammatory pathway regulates innate and adaptive immunity, and modulation of this reflex by vagus nerve stimulation (VNS) is effective in various inflammatory disease models, such as rheumatoid arthritis and inflammatory bowel disease. Effectiveness of VNS in these models necessitates the integration of neural signals and α7 nicotinic acetylcholine receptors (α7nAChRs) on splenic macrophages. Here, we sought to determine whether electrical stimulation of the vagus nerve attenuates kidney ischemia-reperfusion injury (IRI), which promotes the release of proinflammatory molecules. Stimulation of vagal afferents or efferents in mice 24 hours before IRI markedly attenuated acute kidney injury (AKI) and decreased plasma TNF. Furthermore, this protection was abolished in animals in which splenectomy was performed 7 days before VNS and IRI. In mice lacking α7nAChR, prior VNS did not prevent IRI. Conversely, adoptive transfer of VNS-conditioned α7nAChR splenocytes conferred protection to recipient mice subjected to IRI. Together, these results demonstrate that VNS-mediated attenuation of AKI and systemic inflammation depends on α7nAChR-positive splenocytes.
Authors
Tsuyoshi Inoue, Chikara Abe, Sun-sang J. Sung, Stefan Moscalu, Jakub Jankowski, Liping Huang, Hong Ye, Diane L. Rosin, Patrice G. Guyenet, Mark D. Okusa
Citation Information: J Clin Invest. 2016. https://doi.org/10.1172/JCI80304.
Abstract
The mTOR pathway orchestrates cellular homeostasis. The rapamycin-sensitive mTOR complex (mTORC1) in the kidney has been widely studied; however, mTORC2 function in renal tubules is poorly characterized. Here, we generated mice lacking mTORC2 in the distal tubule (
Authors
Florian Grahammer, Viatcheslav Nesterov, Azaz Ahmed, Frederic Steinhardt, Lukas Sandner, Frederic Arnold, Tomke Cordts, Silvio Negrea, Marko Bertog, Marcus A. Ruegg, Michael N. Hall, Gerd Walz, Christoph Korbmacher, Ferruh Artunc, Tobias B. Huber
Citation Information: J Clin Invest. 2016. https://doi.org/10.1172/JCI74997.
Abstract
The adult kidney plays a central role in erythropoiesis and is the main source of erythropoietin (EPO), an oxygen-sensitive glycoprotein that is essential for red blood cell production. Decreases of renal pO2 promote hypoxia-inducible factor 2–mediated (HIF-2–mediated) induction of EPO in peritubular interstitial fibroblast-like cells, which serve as the cellular site of EPO synthesis in the kidney. It is not clear whether HIF signaling in other renal cell types also contributes to the regulation of EPO production. Here, we used a genetic approach in mice to investigate the role of renal epithelial HIF in erythropoiesis. Specifically, we found that HIF activation in the proximal nephron via induced inactivation of the von Hippel–Lindau tumor suppressor, which targets the HIF-α subunit for proteasomal degradation, led to rapid development of hypoproliferative anemia that was associated with a reduction in the number of EPO-producing renal interstitial cells. Moreover, suppression of renal EPO production was associated with increased glucose uptake, enhanced glycolysis, reduced mitochondrial mass, diminished O2 consumption, and elevated renal tissue pO2. Our genetic analysis suggests that tubulointerstitial cellular crosstalk modulates renal EPO production under conditions of epithelial HIF activation in the kidney.
Authors
Navid M. Farsijani, Qingdu Liu, Hanako Kobayashi, Olena Davidoff, Feng Sha, Joachim Fandrey, T. Alp Ikizler, Paul M. O’Connor, Volker H. Haase
Citation Information: J Clin Invest. 2016. https://doi.org/10.1172/JCI82592.
Abstract
Focal segmental glomerulosclerosis (FSGS) is a syndrome that involves kidney podocyte dysfunction and causes chronic kidney disease. Multiple factors including chemical toxicity, inflammation, and infection underlie FSGS; however, highly penetrant disease genes have been identified in a small fraction of patients with a family history of FSGS. Variants of apolipoprotein L1 (
Authors
Haiyang Yu, Mykyta Artomov, Sebastian Brähler, M. Christine Stander, Ghaidan Shamsan, Matthew G. Sampson, J. Michael White, Matthias Kretzler, Jeffrey H. Miner, Sanjay Jain, Cheryl A. Winkler, Robi D. Mitra, Jeffrey B. Kopp, Mark J. Daly, Andrey S. Shaw



Copyright © 2024 American Society for Clinical Investigation
ISSN: 0021-9738 (print), 1558-8238 (online)